Life Sciences
· ID-Flow 3, 5, 6 and 9
4
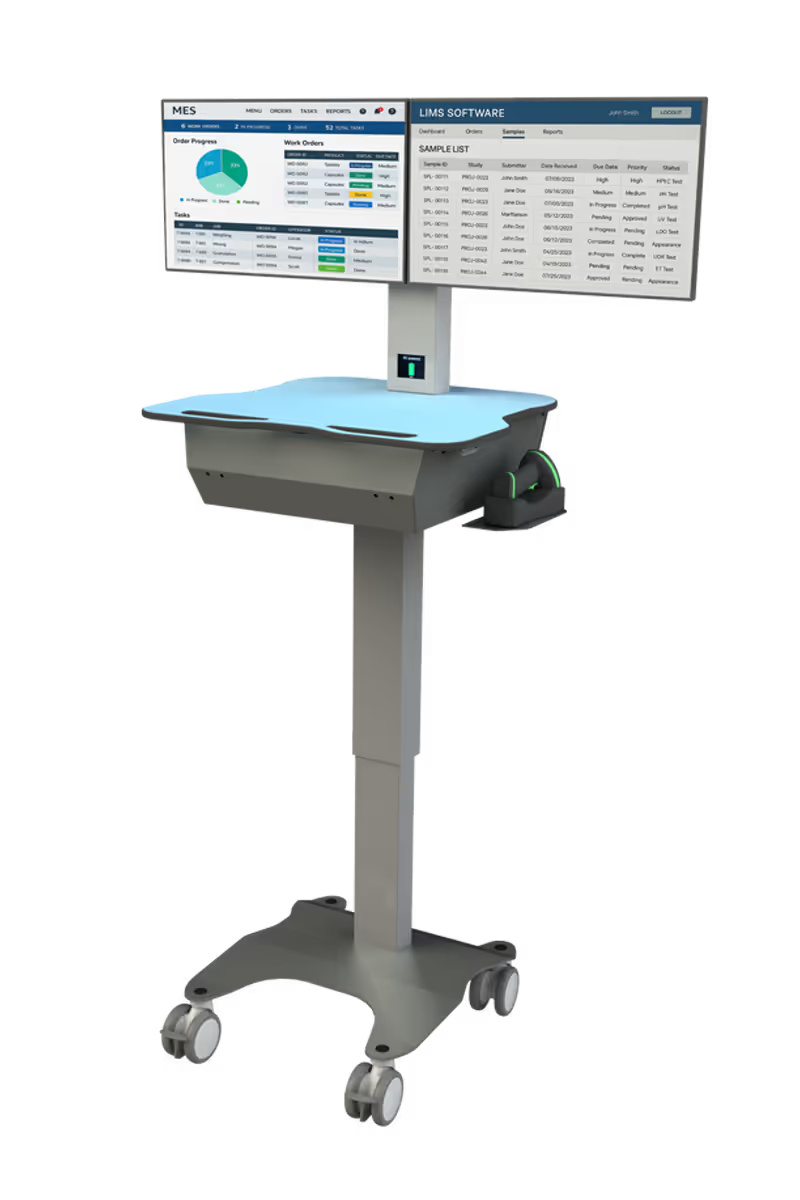
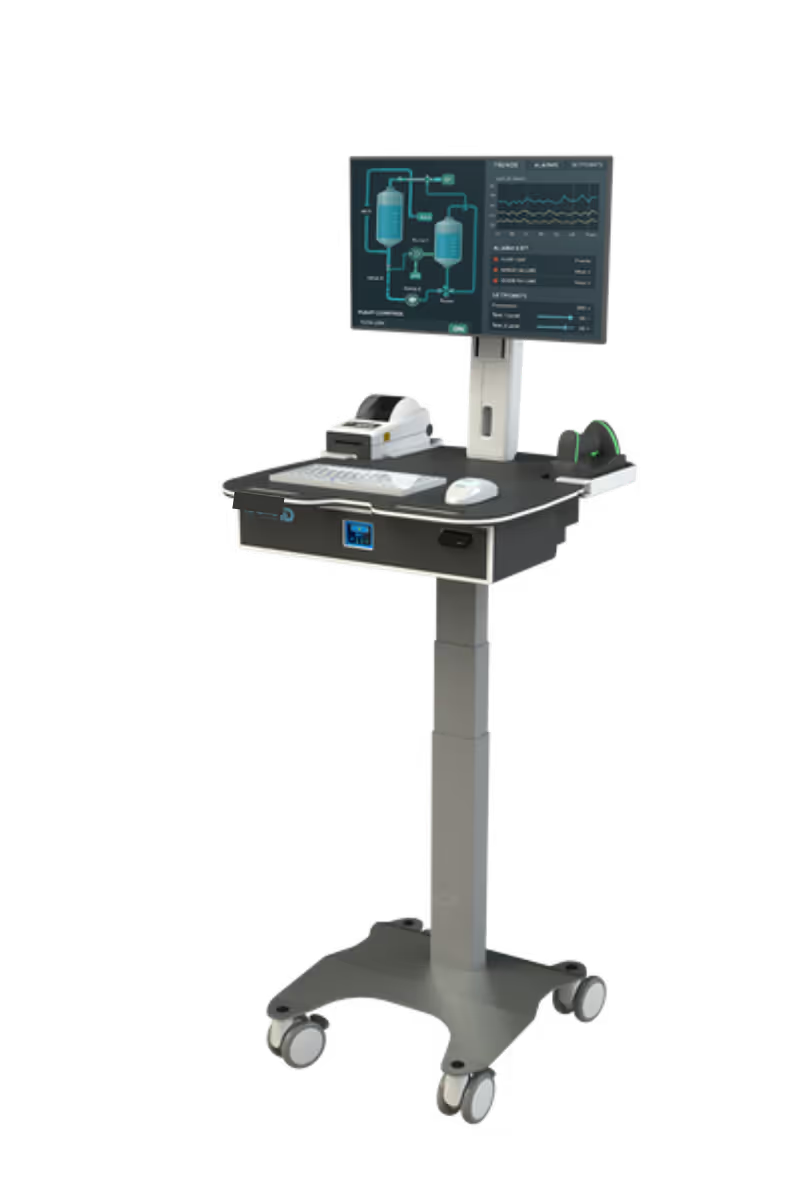
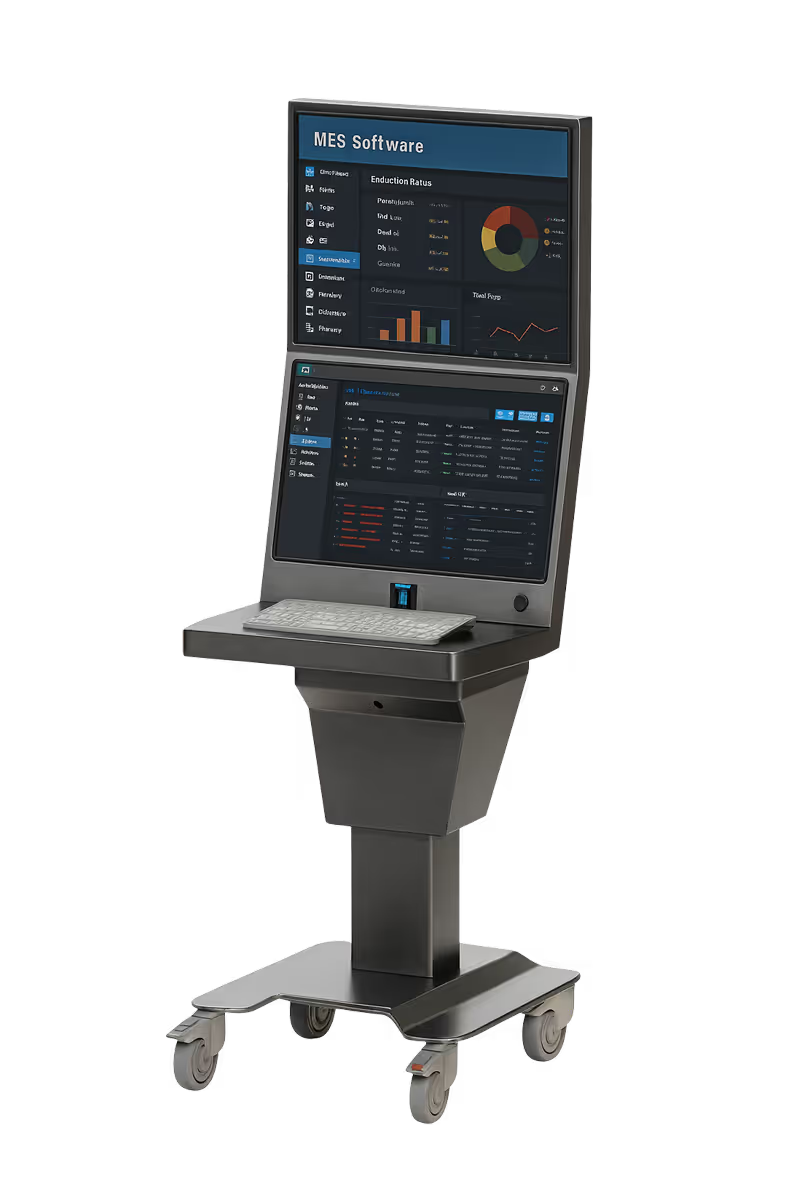
ID-Flow variants deployed across US life sciences sites
316L
fully sealed watertight stainless developed for this customer
DeltaV
+ Syncade · Emerson MES and DCS integration
With Kinetic-ID, you get exactly what you want, and they will focus it to your needs on a practical level.